Chapterwise Solution Particulate Nature of Matter
Question 1
Choose the correct option.
The primary difference between solids and liquids is that the constituent particles are:
- closely packed in solids, while they are stationary in liquids.
- far apart in solids and have fixed position in liquids.
- always moving in solids and have fixed position in liquids.
- closely packed in solids and move past each other in liquids.
Answer: iV. closely packed in solids and move past each other in liquids. Chapterwise Solution Particulate Nature of Matter
Explanation:
- In solids, particles are tightly packed and vibrate in fixed positions.
- In liquids, particles are still close but can slide past one another, allowing flow.
Question 2
Which of the following statements are true? Correct the false statements. Chapterwise Solution Particulate Nature of Matter
- Melting ice into water is an example of the transformation of a solid into a liquid.
- Melting process involves a decrease in interparticle attractions during the transformation.
- Solids have a fixed shape and a fixed volume.
- The interparticle interactions in solids are very small.
- When we heat camphor in one corner of a room, the fragrance reaches all corners of the room.
- On heating, we are adding energy to the camphor, and the energy is released as a smell.
Answer:
| Statement | True/False | Correction (if false) |
| (i) Melting ice into water is an example of the transformation of a solid into a liquid. | True | — |
| (ii) Melting process involves a decrease in interparticle attractions during the transformation. | True | — |
| (iii) Solids have a fixed shape and a fixed volume. | True | — |
| (iv) The interparticle interactions in solids are very strong, and the interparticle spaces are very small. | True | — |
| (v) When we heat camphor in one corner of a room, the fragrance reaches all corners of the room. | True | — |
| (vi) On heating, we are adding energy to the camphor, and the energy is released as a smell. | False | Correction: On heating, camphor undergoes sublimation. The particles disperse in air and reach our nose as smell—not energy being released as smell. |
Question 3
Choose the correct answer with justification. If we could remove all the constituent particles from a chair, what would happen? Chapterwise Solution Particulate Nature of Matter
- Nothing will change. Chapterwise Solution Particulate Nature of Matter
- The chair will weigh less due to lost particles. Chapterwise Solution Particulate Nature of Matter
- Nothing of the chair will remain. Chapterwise Solution Particulate Nature of Matter
Answer: (III). Nothing of the chair will remain.
Justification:
- A chair is made of matter, which consists of particles.
- If all particles are removed, no matter remains—hence, the chair ceases to exist.
Question 4
Why do gases mix easily, while solids do not? Chapterwise Solution Particulate Nature of Matter
Answer:
Gases mix easily because:
- Their particles are far apart and move freely in all directions.
- There are very weak interparticle forces, allowing particles to spread and intermingle.
- This random motion leads to quick diffusion, even without stirring.
In contrast, solids:
Cannot move freely, so intermixing is not possible without external force (like grinding).
Have tightly packed particles in fixed positions.
Summary Table
| Property | Gases | Solids |
| Particle spacing | Far apart | Tightly packed |
| Particle movement | Free, random motion | Fixed position, slight vibration |
| Mixing behavior | Mix easily via diffusion | Do not mix without force |
| Energy required | None (natural mixing) | High (mechanical or thermal) |
Question 5
When spilled on the table, milk in a glass tumbler, flows and spreads out, but the glass tumbler stays in the same shape. Justify this statement.
Answer:
- Milk is a liquid: Its particles are loosely packed and can slide past each other. This allows it to flow and take the shape of the surface it’s on.
- Glass tumbler is a solid: Its particles are tightly packed in fixed positions. This gives it a definite shape and volume, which does not change when placed on a surface.
This demonstrates the difference in particle arrangement and mobility between solids and liquids.
Justification Summary
- Milk flows and spreads because it is a liquid with loosely held, mobile particles.
- The glass tumbler retains its shape because it is a solid with rigid, tightly packed particles.
Question 6
Represent diagrammatically the changes in the arrangement of particles as ice melts and transforms into water vapour.
Answer:
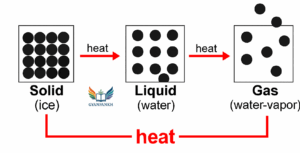
Question 7
Draw a picture representing particles present in the following:
- Aluminium foil
- Glycerin
- Methane gas
Answer:
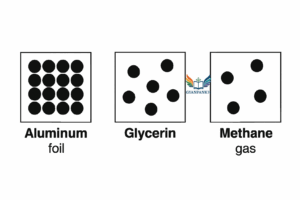
Explanation:
- Particles are tightly packed in a fixed, orderly lattice. They vibrate in place but do not move freely. This structure gives aluminium its rigidity and definite shape. Chapterwise Solution Particulate Nature of Matter
- Particles are closely packed but not fixed. They can slide past each other, allowing glycerin to flow. The arrangement is random, not as rigid as a solid. Chapterwise Solution Particulate Nature of Matter
- Particles are widely spaced and move freely and rapidly. The diagram shows these molecules floating independently, representing the gaseous state. Chapterwise Solution Particulate Nature of Matter
Question 8
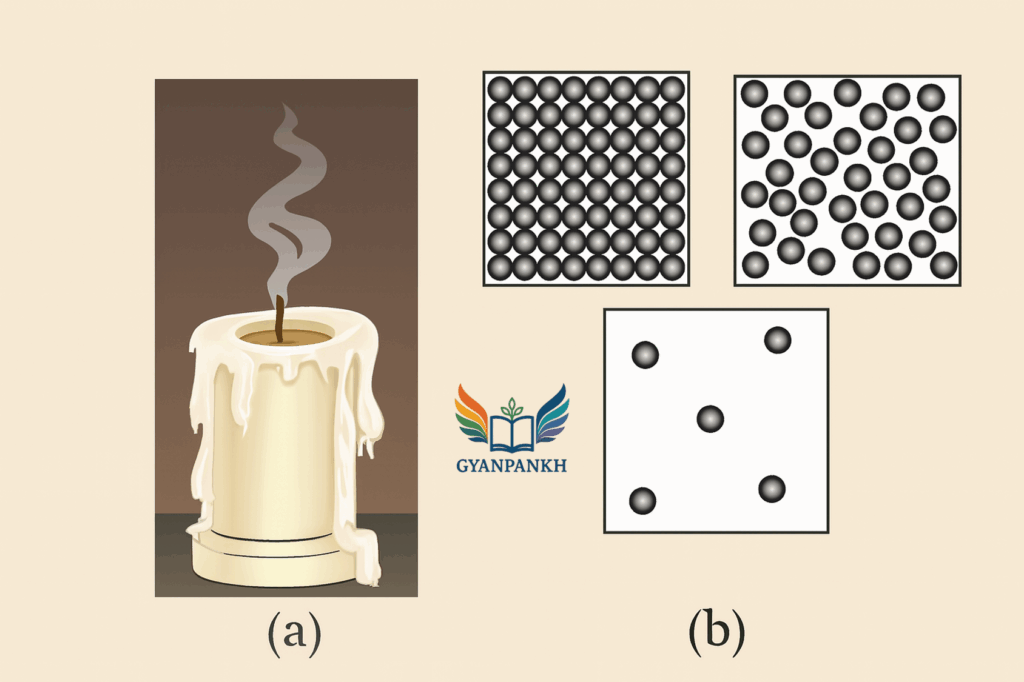
Observe figure A, which shows the image of a candle that was just extinguished after burning for some time. Identify the different states of wax in the figure and match them with Figure B showing the arrangement of particles.
Answer:
When a candle burns and is then extinguished, wax appears in three different states:
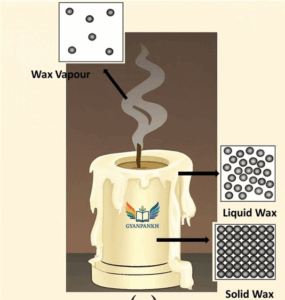
Explanation:
| State of Wax | Observed in Figure A | Particle Arrangement (Figure B) | Key Features |
|---|---|---|---|
| Solid | Unmelted candle body | Top left (tightly packed) | Fixed shape and volume |
| Liquid | Melted wax dripping | Top right (loosely packed) | Flows, takes shape of surface |
| Gas | Smoke rising from wick | Bottom (widely spaced) | Spreads in air, no fixed shape/volume |
Question 9
Why does the water in the ocean taste salty, even though the salt is not visible? Explain.
Answer: Ocean water tastes salty because salt is dissolved in it. When salt dissolves, its particles mix completely with water and become invisible to the eye. However, the salty taste remains because the dissolved salt particles are still present in the water.
Summary
| Feature | Explanation |
| Salt visibility | Not visible because it’s dissolved |
| Taste | Salty due to dissolved sodium and chloride ions |
| Source of salt | Erosion, river runoff, and underwater processes |
Question 10
Grains of rice and rice flour take the shape of the container when placed in different jars. Are they solids or liquids? Explain.
Answer: Grains of rice and rice flour are solids, even though they take the shape of the container. This happens because they are made of tiny solid particles. Each grain or particle keeps its own shape and does not flow like a liquid. They only appear to fill the container because they are small and loosely packed.
Summary
| Material | State of Matter | Reason |
| Rice grains | Solid | Each grain has a fixed shape and volume |
| Rice flour | Solid | Made of tiny solid particles that settle and pile up |
| Water or milk | Liquid | Particles move freely and take the exact shape of the container |
Click here for any Help, Click here for any Suggestions.
