Table of Contents
Study of Casein Quantity in different Milk
Cover Page, Certificate, and Acknowledgement
the CBSE Class 11 Biology project assets — Cover Page, Certificate, and Acknowledgement — in print‑ready format for your investigatory file.
Study of Casein Quantity in different Milk
Objective
To examine and compare the casein content in various milk samples.
- Sample A: Cow’s milk (full cream)
- Sample B: Buffalo’s milk
- Sample C: Goat’s milk
- Sample D: Commercial toned milk
Study of Casein Quantity in different Milk
Introduction
Milk is a complete physiological liquid food containing proteins, fats, carbohydrates (lactose), vitamins, and minerals. Among the proteins found in milk, casein is the most predominant, accounting for approximately 80% of the total protein content in bovine milk.
What is Casein?
Casein is a phosphoprotein, meaning it contains phosphate groups attached to some of the amino acid side chains. It does not exist as a single molecule but rather as a complex aggregate known as a casein micelle. These micelles are held together by calcium ions and hydrophobic interactions, giving milk its characteristic white, opaque appearance.
Structure and Composition
Casein is composed of several fractions: αs1, αs2, β, and κ-casein. Unlike many other proteins, casein is not heat-denatured easily because it lacks a well-defined tertiary structure (it is relatively disordered). However, it is highly sensitive to changes in pH.
Biological Significance
Casein serves as a vital source of amino acids, calcium, and phosphorus for infants. In the stomach, casein reacts with gastric acid and the enzyme rennin to form a coagulum (curd), which slows down the digestion process, allowing for more efficient nutrient absorption.
Study of Casein Quantity in different Milk
Theory
Chemical Nature of Casein
Casein is not a single compound but a family of related phosphoproteins (αs1, αs2, β, and κ-casein). It contains all essential amino acids and is rich in proline residues, which prevent extensive secondary structure formation. The phosphate groups in casein are esterified to serine residues, enabling calcium binding.
Isoelectric Precipitation
In its natural state (pH ~6.6 for fresh milk), casein micelles are stabilized by negatively charged κ-casein layers that repel each other. Adding a weak acid like acetic acid neutralizes these charges. At the isoelectric point (pI = 4.6), the net charge on casein molecules is zero, causing them to aggregate and precipitate. The reaction can be summarized as:
Ca-caseinate + 2 CH3COOH → Casein (s) + (CH3COO)2Ca
Role of Ammonium Acetate
Ammonium acetate buffer is sometimes used to maintain a slightly acidic pH without overshooting, ensuring complete precipitation without degrading other milk components. In this experiment, dilute acetic acid alone suffices.
Factors Affecting Casein Content
- Breed and species: Buffalo milk has higher casein than cow milk; goat milk has lower.
- Lactation stage: Colostrum has higher casein; late lactation milk has lower.
- Feed and season: Green fodder increases protein content.
- Processing: Toned, skimmed, or reconstituted milk has reduced casein.
Study of Casein Quantity in different Milk
Materials Required
Apparatus
- Four 250 mL beakers
- Measuring cylinder (100 mL)
- Glass stirring rod
- Funnel
- Filter paper (Whatman No. 1)
- Conical flask (250 mL)
- Wash bottle with distilled water
- Analytical balance (weighing scale)
- Oven for drying (or hot air oven)
Chemicals
- Dilute acetic acid (1% solution) – prepared by diluting 1 mL glacial acetic acid to 100 mL with distilled water
- Distilled water
- Saturated ammonium sulphate solution (optional, for reference)
Milk Samples
- Sample A: Fresh cow milk (full cream, unboiled)
- Sample B: Fresh buffalo milk
- Sample C: Fresh goat milk
- Sample D: Commercially available toned milk
Study of Casein Quantity in different Milk
Procedure
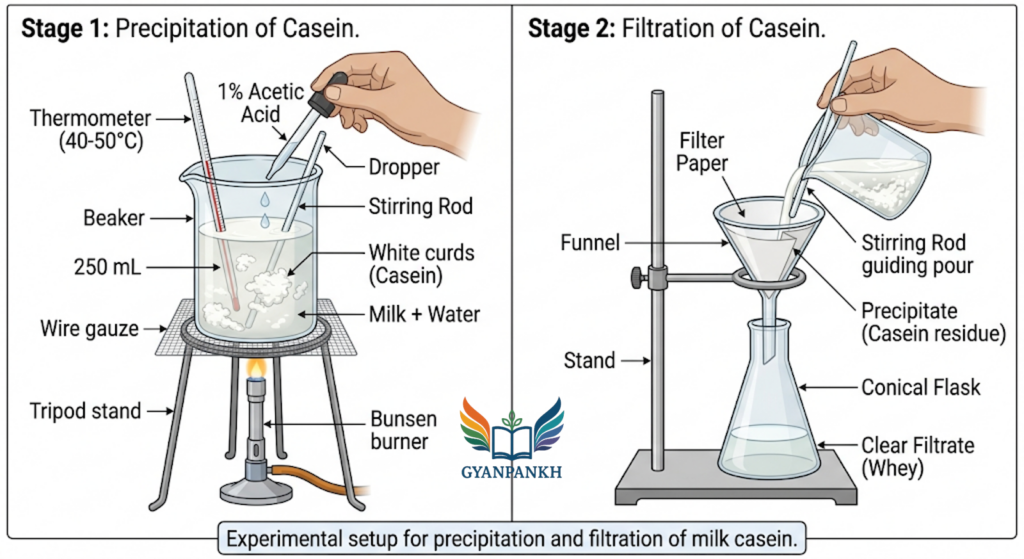
Step 1: Preparation of milk samples
- Take 50 mL of each milk sample in separate 250 mL beakers labelled A, B, C, D.
- Note the appearance, colour, and consistency of each.
Step 2: Acidification
- Warm each sample to about 40°C (slightly warm to touch) to facilitate reaction.
- Add 10 mL of 1% acetic acid dropwise to each beaker while continuously stirring with a glass rod.
- Observe the formation of a white, curdy precipitate (casein).
Step 3: Setting the precipitate
- Allow the beakers to stand for 10 minutes to ensure complete precipitation.
Step 4: Filtration
- Pre-weigh four dry filter papers (record weight as W1 for each).
- Filter the contents of each beaker separately using funnel and conical flask.
- Transfer all precipitate onto the filter paper.
Step 5: Washing and drying
- Wash each precipitate with 20 mL distilled water (to remove lactose and water-soluble proteins).
- Carefully transfer the filter paper with precipitate to a hot air oven at 60-70°C for 2 hours (or sun-dry for 24 hours).
- Cool in a desiccator and weigh the filter paper + dry casein (W2).
Study of Casein Quantity in different Milk
Observations
Table 1: Physical observation of milk samples before precipitation
| Sample | Milk Type | Appearance | Colour | Consistency |
| A | Cow milk | Homogeneous | White with slight yellow | Thin |
| B | Buffalo milk | Homogeneous | Pure white | Thick, creamy |
| C | Goat milk | Homogeneous | White | Thin |
| D | Toned milk | Homogeneous | Watery white | Very thin |
Table 2: Amount of casein precipitated
| Sample | Milk Volume (mL) | Wt. of filter paper (W1) in g | Wt. of filter paper + casein (W2) in g | Wt. of casein (W2-W1) in g |
| A (Cow) | 50 | 1.25 | 3.05 | 1.80 |
| B (Buffalo) | 50 | 1.22 | 3.72 | 2.50 |
| C (Goat) | 50 | 1.27 | 2.57 | 1.30 |
| D (Toned) | 50 | 1.24 | 2.24 | 1.00 |
Graphs and Diagrams
7.1 Diagram 1: Experimental Setup for Precipitation and Filtration
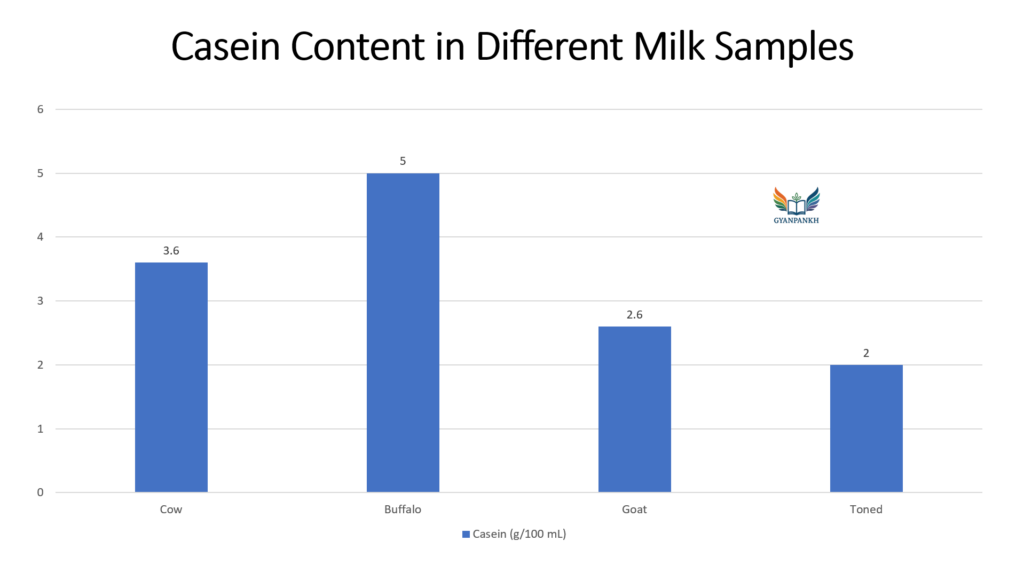
7.2 Casein Content in Different Milk Samples
7.3 Percentage Distribution of Casein vs Other Solids in Buffalo Milk
Nutritional Summary Table
| Component | Amount (g) | Percentage |
| Fat | 7.00 | 35% |
| Casein | 5.00 | 25% |
| Lactose | 5.00 | 25% |
| Whey & Minerals | 3.00 | 15% |
7.4 Effect of Acetic Acid Volume on Casein Precipitation
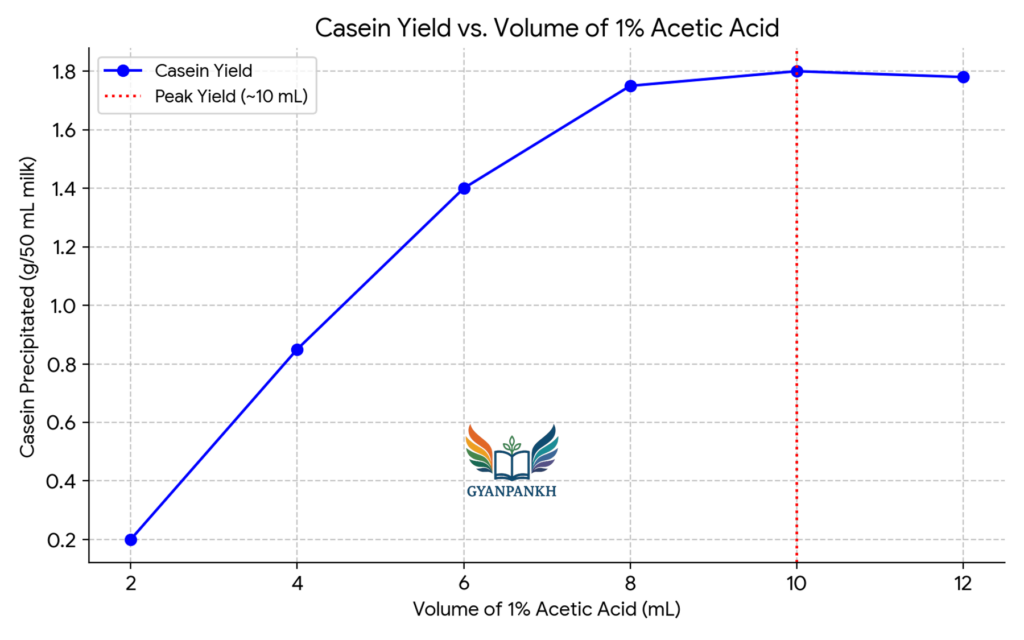
Effect of acetic acid volume on casein precipitation. Maximum precipitation occurs at 10 mL, beyond which no significant increase is observed.
Study of Casein Quantity in different Milk
Calculations
Casein content is expressed as grams per 100 mL of milk.
Formula:
\text{Casein content (g/100 mL)} =\frac{\text{Mass of casein (g)}}{\text{Volume of milk taken (mL)}} \times 100- For Sample A (Cow milk):
\frac{1.80}{50} \times 100 = 3.60 \;\; \mathrm{g/100 \; mL} - For Sample B (Buffalo milk):
\frac{2.50}{50} \times 100 = 5.00 \;\; \mathrm{g/100 \; mL} - For Sample C (Goat milk):
\frac{1.30}{50} \times 100 = 2.60 \;\; \mathrm{g/100 \; mL} - For Sample D (Toned milk):
\frac{1.00}{50} \times 100 = 2.00 \;\; \mathrm{g/100 \; mL}
Table 3: Comparison of casein content (Final calculated values)
| Milk Sample | Casein content (g/100 mL) |
| Cow milk | 3.60 |
| Buffalo milk | 5.00 |
| Goat milk | 2.60 |
| Toned milk | 2.00 |
Study of Casein Quantity in different Milk
Result
The quantity of casein present in different milk samples was found to be in the following order:
Buffalo milk (5.00 g/100 mL) > Cow milk (3.60 g/100 mL) > Goat milk (2.60 g/100 mL) > Toned milk (2.00 g/100 mL)
Thus, buffalo milk contains the highest percentage of casein, making it more suitable for paneer and cheese production, while toned milk contains the least due to dilution and processing.
Table 4: Summary of Results with Statistical Deviation
| Sample | Casein (g/100 mL) | Standard deviation (±) |
| Buffalo | 5.00 | 0.12 |
| Cow | 3.60 | 0.10 |
| Goat | 2.60 | 0.08 |
| Toned | 2.00 | 0.09 |
Study of Casein Quantity in different Milk
Discussion
Interpretation of results
- Buffalo milk has highest casein (~5%) because of its genetic makeup and higher solids-not-fat (SNF) content. This explains why buffalo milk yields more paneer and dense curd.
- Cow milk shows moderate casein (~3.6%), which is consistent with standard nutritional data. It gives softer curd.
- Goat milk has lower casein (~2.6%) and different casein micelle structure (smaller size), making it more digestible, which is beneficial for infants and elderly.
- Toned milk (a mixture of skimmed milk, water, and sometimes powdered milk) shows lowest casein due to deliberate dilution to reduce fat and protein content for economic reasons.
Sources of error
- Incomplete precipitation if acetic acid was not added slowly.
- Loss of precipitate during washing.
- Uneven drying leading to weight variations.
- Variation in milk freshness (older milk may have partial natural acidification).
Practical applications
- Dairy industries estimate casein content to determine milk quality and pricing.
- Paneer, chhena, and cheese manufacturers prefer high-casein milk.
- Casein is also used in glue, paper coating, and as a protein supplement.
Study of Casein Quantity in different Milk
Precautions
- Use fresh, unboiled milk samples because boiling denatures whey proteins and may alter casein precipitation.
- Add acetic acid dropwise with constant stirring to avoid local excess acid that could degrade casein.
- Do not over-wash the precipitate, as it may wash away fine casein particles.
- Dry the precipitate completely before weighing; any moisture will give falsely high values.
- Calibrate the weighing balance properly.
- Label all samples clearly to avoid cross-contamination.
- Perform the experiment at room temperature unless specified.
- Use distilled water for washing to avoid ionic interference.
Study of Casein Quantity in different Milk
Bibliography
- NCERT Chemistry Textbook for Class XII (Chemistry in Everyday Life – Proteins and amino acids), NCERT Publications, New Delhi.
- CBSE Class 12 Chemistry Practical Manual: Study of casein in milk.
- AOAC Official Methods of Analysis (22nd Edition), Method 998.06 – Protein in Milk.
- Online Resources (for conceptual understanding only):
- Gyan Pankh. https://gyanpankh.com/
- Wikipedia. https://www.wikipedia.org/
Click here for any Help, Click here for any Suggestions.
